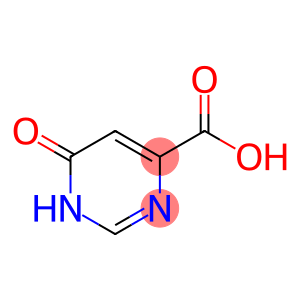
4-Pyrimidinecarboxylicacid, 1,6-dihydro-6-oxo-
6-Hydroxy-4-pyrimidinecarboxylic acid
CAS: 6299-87-2
Molecular Formula: C5H4N2O3
4-Pyrimidinecarboxylicacid, 1,6-dihydro-6-oxo- - Names and Identifiers
4-Pyrimidinecarboxylicacid, 1,6-dihydro-6-oxo- - Physico-chemical Properties
| Molecular Formula | C5H4N2O3 |
| Molar Mass | 140.1 |
| Density | 1.63±0.1 g/cm3(Predicted) |
| Melting Point | 268-270 °C |
| Boling Point | 305.2±52.0 °C(Predicted) |
| Flash Point | 138.4°C |
| Vapor Presure | 0.000191mmHg at 25°C |
| Appearance | Purple powder |
| Color | White to Light yellow to Light red |
| pKa | 2.77±0.20(Predicted) |
| Storage Condition | 2-8°C |
| Refractive Index | 1.662 |
| MDL | MFCD00023267 |
4-Pyrimidinecarboxylicacid, 1,6-dihydro-6-oxo- - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Safety Description | 24/25 - Avoid contact with skin and eyes. |
| HS Code | 29335990 |
4-Pyrimidinecarboxylicacid, 1,6-dihydro-6-oxo- - Reference Information
| Application | as the main formic acid derivative, at the same time, it overcomes the irritation and corrosion of formic acid, and is widely used in feed and aquaculture. As a relatively simple organic acid, 6-hydroxy-4-pyrimidinecarboxylic acid has good acidification and nutritional functions. |
| synthetic method | in a 1 L round-bottom flask, add sodium hydroxide solution (10g) to 500 mL of water 55 g of sodium ethyl oxalate (1.05 eq,0.26 mol) and 26 g of formamidine acetate (1 eq,0.25 mol) were added. The reaction mixture was stirred at room temperature for 16 hours. Concentrated HCl was carefully added to the mixture until pH = 1 and a fine solid precipitated, and the reaction mixture was stirred at 0°C for 1 hour. The solid was filtered and then washed with water and ether. The white solid was then left in a vacuum oven heated at 40°C for 20 hours. Tri-saturation reaction in methanol gave 25% yield of the title compound 6-hydroxy-4-pyrimidinecarboxylic acid. Proton nuclear magnetic resonance spectrum (d6 DMSO):12.88(1H,OH),8.24(s,1H),6.83(s,2H). Figure 6-hydroxy-4-pyrimidinecarboxylic acid synthesis |
Last Update:2024-04-09 02:00:10
Supplier List
Product Name: 6-Hydroxy-4-pyrimidinecarboxylic acid Request for quotation
CAS: 6299-87-2
Tel: 13247110337
Email: 2205437118@qq.com
Mobile: 13247110337
QQ: 2205437118
Wechat: 13247110337
CAS: 6299-87-2
Tel: 13247110337
Email: 2205437118@qq.com
Mobile: 13247110337
QQ: 2205437118
Wechat: 13247110337
Spot supply
Product Name: 6-hydroxypyrimidine-4-carboxylic acid Visit Supplier Webpage Request for quotationCAS: 6299-87-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 6-Hydroxy-4-pyrimidinecarboxylic acid Request for quotation
CAS: 6299-87-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 6299-87-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 6-Oxo-3H-pyrimidine-4-carboxylic acid Request for quotation
CAS: 6299-87-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 6299-87-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 6-Oxo-3H-Pyrimidine-4-Carboxylic Acid Visit Supplier Webpage Request for quotation
CAS: 6299-87-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 6299-87-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 6-Hydroxy-4-pyrimidinecarboxylic acid Request for quotation
CAS: 6299-87-2
Tel: 13247110337
Email: 2205437118@qq.com
Mobile: 13247110337
QQ: 2205437118
Wechat: 13247110337
CAS: 6299-87-2
Tel: 13247110337
Email: 2205437118@qq.com
Mobile: 13247110337
QQ: 2205437118
Wechat: 13247110337
Spot supply
Product Name: 6-hydroxypyrimidine-4-carboxylic acid Visit Supplier Webpage Request for quotationCAS: 6299-87-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 6-Hydroxy-4-pyrimidinecarboxylic acid Request for quotation
CAS: 6299-87-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 6299-87-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 6-Oxo-3H-pyrimidine-4-carboxylic acid Request for quotation
CAS: 6299-87-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 6299-87-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 6-Oxo-3H-Pyrimidine-4-Carboxylic Acid Visit Supplier Webpage Request for quotation
CAS: 6299-87-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 6299-87-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




